|
2/29/2024 0 Comments Lithium nmc cathode
Electron microscopy studies indicated that, in lithium and manganese-rich materials, the capacity fading and voltage decay were partially attributed to structural reconstruction inducing a transition from the layered structure to spinel and/or rock-salt structures at the surface and/or in the bulk 5, 17, 25, 26. Due to the structural and chemical complexities of these layered structures, their fading mechanisms have not been fully resolved. With respect to regular NMC materials, although improved high-voltage cyclability (2.0–4.7 V versus Li +/Li) was recently achieved when a small percentage of Co was substituted with Ti (<4%), capacity fading still occurred after extended charge–discharge cycles 18. Specifically, high-voltage charge capacities achieved in lithium-rich/manganese-rich layered cathodes are directly associated with various irreversible electrochemical processes including oxygen loss and concomitant lithium ion removal 23 and electrode/electrolyte reactions 24. Practical implementation of some of these materials is thwarted by their high first-cycle coulombic inefficiencies 17, 18, 19, 20, capacity fading 18, 21 and voltage instability 20, 21, 22, especially during high-voltage operation. (1−x)LiMO 2 (M=Mn, Ni, Co, and so on) materials, is technologically significant as they represent a group of materials with the potential to improve energy densities and reduce costs for plug-in hybrid electric vehicles and electric vehicles 14, 15, 16, 17.The investigation of in situ surface reconstruction of layered cathode materials, such as stoichiometric LiNi xMn xCo 1−2xO 2 (that is, NMC), lithium-rich Li(Li yNi x−yMn xCo 1−2x)O 2, lithium-rich/manganese-rich (composite layered–layered) xLi 2MnO 3 An improved understanding of in situ surface reconstruction phenomena imparts knowledge not only for understanding degradation mechanisms for battery electrodes but also to provide insights into the surface functionalization for enhanced cyclability 12, 13. 
Specifically, the surfaces of lithium-ion battery electrodes evolve simultaneously with charge–discharge cycling (that is, in situ surface reconstruction and formation of a surface reaction layer (SRL)) that can lead to deterioration of performance 4, 5, 11. Structural and/or chemical rearrangements at surfaces determine the way a material interacts with its surrounding environment, thus controlling the functionalities of the material 6, 7, 8, 9, 10. This work sets a refined example for the study of surface reconstruction and chemical evolution in battery materials using combined diagnostic tools at complementary length scales.Ĭhemical evolution and structural transformations at the surface of a material directly influence characteristics relevant to a wide range of prominent applications including heterogeneous catalysis 1, 2, 3 and energy storage 4, 5. Furthermore, the surface reaction layer is composed of lithium fluoride embedded in a complex organic matrix. It was found that the surface reconstruction exhibits a strong anisotropic characteristic, which predominantly occurs along lithium diffusion channels. These are primarily responsible for the prevailing capacity fading and impedance buildup under high-voltage cycling conditions, as well as the first-cycle coulombic inefficiency. Using correlated ensemble-averaged high-throughput X-ray absorption spectroscopy and spatially resolved electron microscopy and spectroscopy, here we report structural reconstruction (formation of a surface reduced layer, to transition) and chemical evolution (formation of a surface reaction layer) at the surface of LiNi xMn xCo 1−2xO 2 particles. 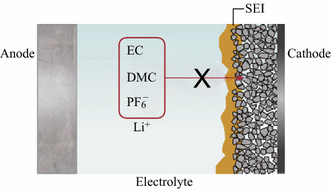
The present study sheds light on the long-standing challenges associated with high-voltage operation of LiNi xMn xCo 1−2xO 2 cathode materials for lithium-ion batteries.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
